How Many Milliliters Is 60 G
Greels
Apr 02, 2025 · 5 min read
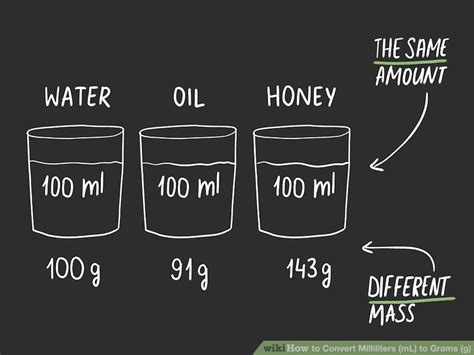
Table of Contents
How Many Milliliters is 60g? Understanding Volume and Mass
Converting between grams (g) and milliliters (ml) isn't a straightforward process like converting between different units of the same measurement (e.g., kilometers to meters). This is because grams measure mass (the amount of matter in an object), while milliliters measure volume (the amount of space an object occupies). The conversion depends entirely on the density of the substance you're measuring. Density is the mass per unit volume, often expressed as grams per milliliter (g/ml) or grams per cubic centimeter (g/cm³). Since 1 ml is equal to 1 cm³, these units are interchangeable in this context.
Let's explore this concept in detail and learn how to perform this conversion accurately for different substances.
The Crucial Role of Density
The key to understanding how many milliliters are in 60g is understanding density. Density is a material-specific property; water, oil, and gold all have different densities. This means that 60g of water will occupy a different volume than 60g of oil or 60g of gold.
The formula connecting mass, volume, and density is:
Density (ρ) = Mass (m) / Volume (V)
We can rearrange this formula to solve for volume:
Volume (V) = Mass (m) / Density (ρ)
This formula is the cornerstone of converting grams to milliliters. To use it, you need to know the density of the substance you're working with.
Density of Common Substances
Here are the densities of some common substances, approximately at room temperature:
- Water: 1 g/ml (This is a very convenient density!)
- Milk: ~1.03 g/ml
- Vegetable Oil: ~0.92 g/ml
- Honey: ~1.42 g/ml
- Ethanol (Alcohol): ~0.79 g/ml
- Mercury: ~13.6 g/ml
- Gold: ~19.3 g/ml
Note: These are approximate values, and the actual density can vary slightly based on factors like temperature and purity.
Calculating Volume for Different Substances
Now let's use the formula to calculate the volume for 60g of different substances:
Example 1: Water
- Mass (m) = 60g
- Density (ρ) = 1 g/ml
Volume (V) = 60g / 1 g/ml = 60 ml
Therefore, 60g of water occupies a volume of 60 ml.
Example 2: Vegetable Oil
- Mass (m) = 60g
- Density (ρ) = 0.92 g/ml
Volume (V) = 60g / 0.92 g/ml ≈ 65.2 ml
Therefore, 60g of vegetable oil occupies a volume of approximately 65.2 ml.
Example 3: Honey
- Mass (m) = 60g
- Density (ρ) = 1.42 g/ml
Volume (V) = 60g / 1.42 g/ml ≈ 42.3 ml
Therefore, 60g of honey occupies a volume of approximately 42.3 ml.
Example 4: Gold
- Mass (m) = 60g
- Density (ρ) = 19.3 g/ml
Volume (V) = 60g / 19.3 g/ml ≈ 3.11 ml
Therefore, 60g of gold occupies a volume of approximately 3.11 ml.
Understanding the Implications
These examples clearly illustrate that the volume occupied by 60g of a substance varies greatly depending on its density. A substance with a high density (like gold) will have a much smaller volume for the same mass compared to a substance with a low density (like vegetable oil).
Beyond Simple Calculations: Factors Affecting Density
While the above examples provide a good understanding of the basic conversion, it's crucial to remember that several factors can influence density:
- Temperature: Density typically decreases as temperature increases (except for water, which has its highest density at 4°C).
- Pressure: Increased pressure generally leads to increased density.
- Purity: Impurities in a substance can alter its density.
- Concentration: For solutions, the concentration of the solute impacts the overall density.
Practical Applications and Considerations
Understanding the relationship between mass, volume, and density has numerous practical applications:
- Cooking and Baking: Recipes often specify ingredients by weight (grams) rather than volume (milliliters) for accuracy. Knowing the density of ingredients helps in conversions.
- Chemistry and Physics: Density is a fundamental property used extensively in scientific calculations and experiments.
- Engineering: Density plays a vital role in structural design and material selection.
- Medicine: Density measurements are used in various diagnostic procedures.
Frequently Asked Questions (FAQs)
Q: Can I use an online converter to find the volume of 60g of a substance?
A: Yes, many online converters are available. However, remember to input the correct density for the specific substance you're working with. The accuracy of the result depends entirely on the accuracy of the density value used.
Q: What if I don't know the density of the substance?
A: If you don't know the density, you will need to determine it experimentally. This typically involves measuring both the mass and the volume of a sample of the substance using appropriate tools like a balance and a graduated cylinder or volumetric flask. Then, you can calculate the density using the formula: Density = Mass / Volume.
Q: Are there any limitations to this conversion method?
A: The accuracy of the conversion is directly linked to the accuracy of the density value used. Furthermore, this conversion is primarily applicable to homogeneous substances (substances with uniform composition throughout). Heterogeneous mixtures might require more complex approaches to determine their average density.
Q: What units are commonly used to express density?
A: Density is commonly expressed in grams per milliliter (g/ml), grams per cubic centimeter (g/cm³), or kilograms per liter (kg/L). These units are all interchangeable.
Conclusion
Converting 60g to milliliters isn't a simple matter of multiplying by a constant factor. It requires knowing the density of the specific substance. By understanding the relationship between mass, volume, and density and using the provided formula, you can accurately convert between grams and milliliters for various substances. Remember to consider the potential influence of factors such as temperature, pressure, and purity on the density of the material. This knowledge is essential for accurate calculations across various fields, from cooking to scientific research and engineering applications.
Latest Posts
Latest Posts
-
What Is 72 In In Feet
Apr 03, 2025
-
1 75 M In Feet And Inches
Apr 03, 2025
-
What Is 130 Cm In Inches
Apr 03, 2025
-
How Much Is 151 Cm In Feet
Apr 03, 2025
-
How Many Pounds Is 25 Ounces
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many Milliliters Is 60 G . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
