60 Grams Equals How Many Milliliters
Greels
Apr 03, 2025 · 4 min read
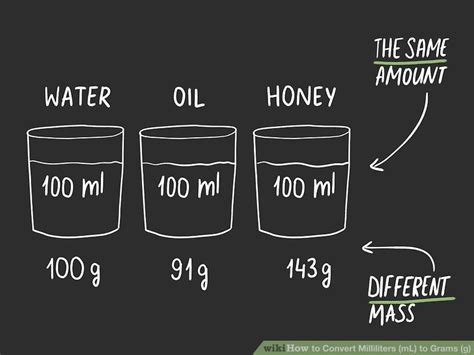
Table of Contents
60 Grams Equals How Many Milliliters? Understanding the Relationship Between Mass and Volume
The question "60 grams equals how many milliliters?" doesn't have a single, straightforward answer. This is because grams (g) measure mass, while milliliters (mL) measure volume. The conversion depends entirely on the density of the substance you're measuring. Density is the mass per unit volume, often expressed as grams per milliliter (g/mL) or grams per cubic centimeter (g/cm³; 1 mL = 1 cm³).
This article will delve deep into understanding the relationship between mass and volume, exploring different scenarios and providing you with the tools to accurately convert grams to milliliters for various substances. We'll also cover related concepts crucial for accurate scientific measurements and practical applications.
Understanding Density: The Key to the Conversion
The fundamental principle connecting mass and volume is density. Density tells us how much mass is packed into a given volume. A denser substance will have more mass in the same volume compared to a less dense substance. The formula for density is:
Density (ρ) = Mass (m) / Volume (V)
To convert grams to milliliters, we need to rearrange this formula:
Volume (V) = Mass (m) / Density (ρ)
Therefore, to find the volume (in milliliters) equivalent to 60 grams, we must know the density of the substance.
Examples: Converting 60 Grams to Milliliters for Different Substances
Let's explore a few examples to illustrate how the density affects the conversion:
1. Water: The Benchmark
Water is often used as a reference point because its density is approximately 1 g/mL at 4°C (39°F). This means 1 gram of water occupies a volume of 1 milliliter. Therefore, for water:
- Volume (V) = 60 g / 1 g/mL = 60 mL
60 grams of water occupies a volume of 60 milliliters.
2. Oil: A Less Dense Substance
Vegetable oil, for instance, has a density slightly less than water, typically around 0.92 g/mL. To convert 60 grams of vegetable oil to milliliters:
- Volume (V) = 60 g / 0.92 g/mL ≈ 65.22 mL
60 grams of vegetable oil occupies a volume of approximately 65.22 milliliters. Notice that it occupies a larger volume than the same mass of water due to its lower density.
3. Mercury: A Dense Substance
Mercury is a much denser liquid than water, with a density of approximately 13.5 g/mL. Let's calculate the volume of 60 grams of mercury:
- Volume (V) = 60 g / 13.5 g/mL ≈ 4.44 mL
60 grams of mercury occupies a volume of approximately 4.44 milliliters. This demonstrates how a denser substance occupies a much smaller volume for the same mass.
4. Solids: The Importance of Shape and Packing
Converting the mass of a solid to volume is more complex because the density of solids can vary depending on their crystalline structure and packing. Furthermore, the shape of the solid affects its volume. For irregularly shaped solids, determining the volume often involves water displacement methods.
Let's consider a hypothetical solid with a density of 2.5 g/mL. The volume of 60 grams of this solid would be:
- Volume (V) = 60 g / 2.5 g/mL = 24 mL
Practical Applications and Considerations
Understanding the relationship between grams and milliliters is essential in numerous fields:
-
Cooking and Baking: Recipes often use volume measurements for liquids. Knowing the density can help you convert between mass and volume for more accurate results.
-
Chemistry and Science: Accurate measurements are crucial in chemical experiments and scientific research. Density is a fundamental property used in many calculations.
-
Medicine and Pharmaceuticals: Dosage calculations frequently involve converting between mass and volume, especially for liquid medications.
-
Engineering and Manufacturing: Density is a key factor in material selection and design processes, particularly in applications involving fluid dynamics or structural integrity.
-
Environmental Science: Density is used to understand the properties of different materials in environmental studies, such as soil density and water quality analysis.
Beyond Simple Conversions: Advanced Concepts
While the basic conversion using the density formula is straightforward, several factors can influence accuracy:
-
Temperature: Density can change with temperature. The densities used in our examples are approximate and can vary slightly depending on the temperature.
-
Pressure: Pressure also influences the density of substances, especially gases.
-
Purity: Impurities in a substance can affect its density. The purity of the substance must be considered for precise conversions.
-
State of Matter: The density of a substance changes significantly depending on whether it is a solid, liquid, or gas.
Conclusion: Mastering Gram-to-Milliliter Conversions
Converting 60 grams to milliliters requires understanding the concept of density and applying the appropriate formula. There's no single answer without knowing the substance's density. This article provides a comprehensive overview of the principles involved, practical examples, and considerations for accurate conversions. Remember that density is a crucial factor, and temperature and purity can influence the results. Mastering this conversion will be valuable in various scientific, culinary, and everyday applications. By understanding the relationship between mass and volume, you can perform accurate measurements and calculations, leading to better results in your work.
Latest Posts
Latest Posts
-
X Square Root Of X 6
Apr 04, 2025
-
69 Kg Is How Many Pounds
Apr 04, 2025
-
How Many Cm Is 36 In
Apr 04, 2025
-
How Big Is 45 Mm In Inches
Apr 04, 2025
-
How Much Is 750 Kg In Pounds
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about 60 Grams Equals How Many Milliliters . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
